Platform
AFFDO
Automated Force Field Development & Optimization

AFFDO
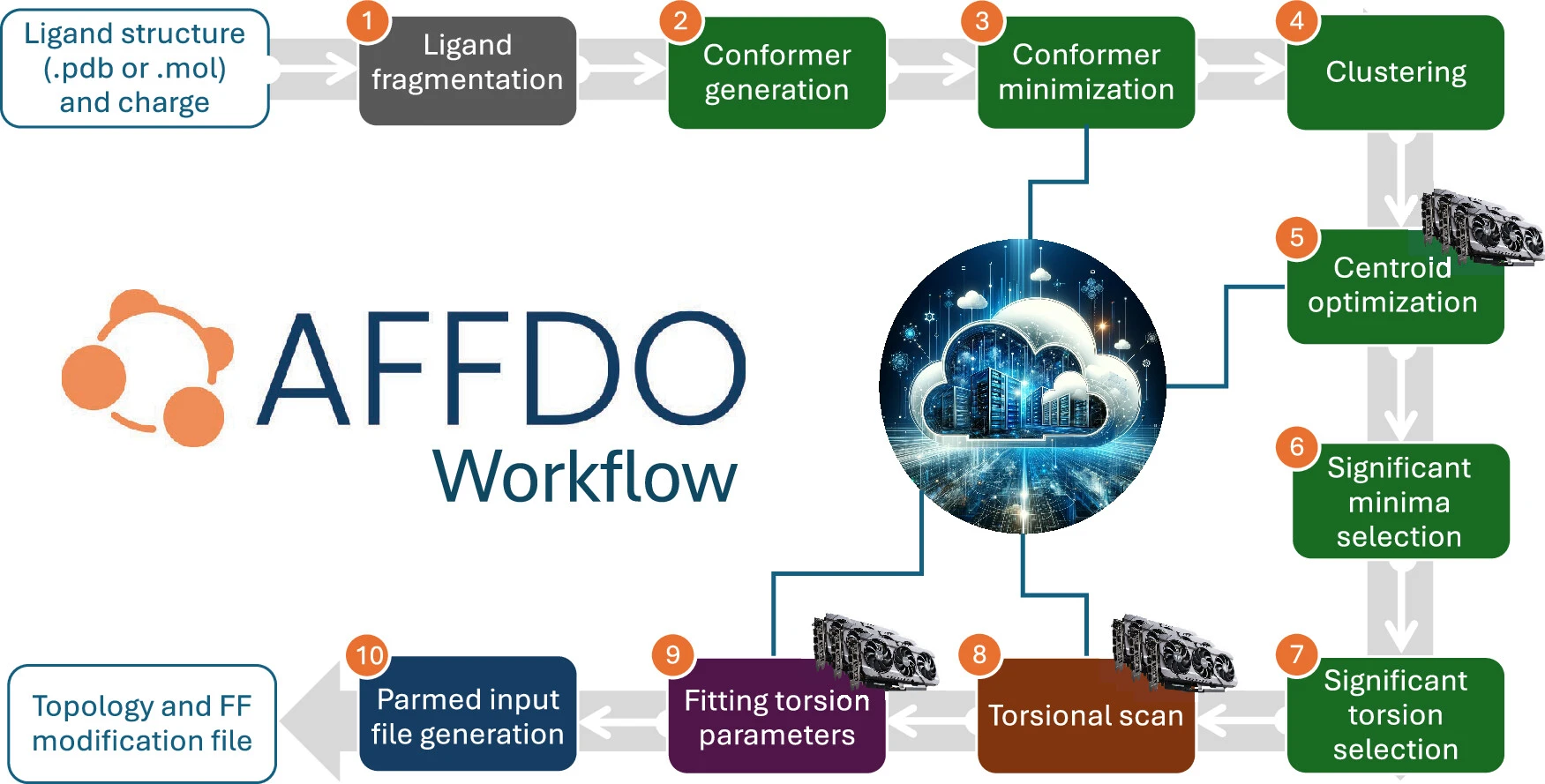
Overview
The use of computer models and simulations for drug discovery has established itself as an important approach to find new molecular entities with desired therapeutic outcomes. Computational tools are now used in all phases in the drug discovery process including the initial screening (hit identification) and further optimization of suitable molecules (lead optimization). Despite the availability of powerful supercomputers and advanced computational tools, the research and development cost of drugs remains extremely high both monetarily (~$6 bn) and time-wise (more than 10 years).
The Challenge
An essential step in lead optimization is predicting the relative binding free energies (RBFE) between the ligand and the target protein, usually done using computational methods such as free energy perturbation (FEP) and thermodynamic integration (TI). The accuracy of these predictions is crucial in identifying candidates with low false-positive rates, especially when the ligand-bound environment undergoes significant changes or if there are electronic and conformational changes that must be properly accounted for.
While quantum mechanical (QM) methods yield highly accurate results, they are far from being directly applicable in RBFE calculations due to their computational costs. Therefore, molecular mechanical (MM) models — classical force fields — constitute the main workhorse in structure-based drug discovery. However, a generic force field lacks the desired specificity for a given protein-ligand target complex. Force field development and optimization are labor-intensive tasks, often dubbed a “black art” due to the expert intuition needed in the design of the model and the generation of a suitable training set.
Our Solution
AFFDO is a novel, flexible and user-friendly computational infrastructure that allows scientists to quickly generate reference training datasets through high-throughput ab initio calculations and transform them into accurate advanced models for RBFE calculations. The platform automates the entire bespoke torsion parameterization workflow — from fragmentation and torsion selection, through reference data generation, to automated fitting — all GPU-accelerated and powered by VCM for scalable cloud orchestration.
Key Features
- Automated & user-friendly — minimal manual intervention required
- Multi-engine pipeline — integrates RDKit, GFN2-xTB, QUICK DFT, and JAX across 10 steps
- GPU-accelerated — reference data generation powered by QUICK DFT on cloud GPUs
- Scalable — powered by VCM for community cloud, AWS, and on-site resources
- Validated accuracy — AFFDO-refined torsions closely match quantum chemistry reference and improve downstream RBFE predictions